- Phylum
- Dinoflagellata
- Class
- Dinophyceae
- Order
- Peridiniales
- Habitat
- plankton, pelagial, littoral
- Distinctive features
- mixotroph. Too large to be grazed by zooplankton. Major food source for Cichlid fishes. Performs daily vertical migrations. When it blooms in spring it constitutes > 95% of total phytoplankton biomass. Its bloom can be seen as patches of coffee brown colored water. Peridinium gatunense is certainly the most studied organism in Lake Kinneret, with hundreds of publications addressing it. For a review article see Zohary et al. 2014.
- Organization
- flagellated single cells
- Color
- dark brown
- Cell shape
- prolate sphaeroid (close to a sphere)
- Cell diameter (D)
- 44 - 58 µm, median: 50 µm (N=1230)
- Cell length (L)
- 47 – 60 µm, median: 52 µm (N=423)
- Cell biovolume
- 44,300 – 99,400 µm³, median: 67,000 µm³.
- Biovolume equation
- V, µm³ = 1.31 D²·⁷⁸³⁵. Regression eqn. based on >400 measurements of both D and L with V computed for a prolate sphaeroid.
Morphological features
This is the second largest dinoflagellate in Lake Kinneret (after Ceratium hirundinella). Under the light microscope, those large, spherical single cells are distinct by having thick, colorless thecae (shell) enveloping the strongly brown-colored living cell or protoplast (Plate 1a,b). The epitheca and hypotheca, are conical and of similar size. The cingulum is wide, deep, and displaced about one cingular width (Plate 2). The sulcus widens slightly and reaches the antapex. The plates are reticulated or rarely rugose and irregularly arranged. Chloroplasts are numerous and parietal (along cell walls). See Hansen and Flaim (2007) for a more detailed taxonomic description. Under unfavorable conditions the cells shed their thecae, and a naked protoplast emerges which will later excrete new thecae. The thecae are also shed before cell division (Plate 1c). After division, each of the two daughter cells excretes its own new thecae. Mitotic cell division is “phased”: it occurs only at night, between midnight and 4 am (Serruya and Pollingher 1976). Sexual reproduction was never observed in Lake Kinneret, possibly because it also occurs at night. Peridinium gatunense resting cysts are spherical, with a thick and distinct cell wall, and two distinct red spots, or eyespots (Plate 3). Throughout the bloom development period, a small proportion of the cells (~ 1 %, Alster et al. 2006) change their morphology to become resting cysts, which sink to the sediments to remain there as an inoculum for future growth. Vegetative cells emerging from resting cysts keep the two red spots. After cell division each daughter cell keeps one red spot. With later divisions those red spots get diluted. Presence of those red spots are thus an indication of the origin of the cell from a resting cyst.
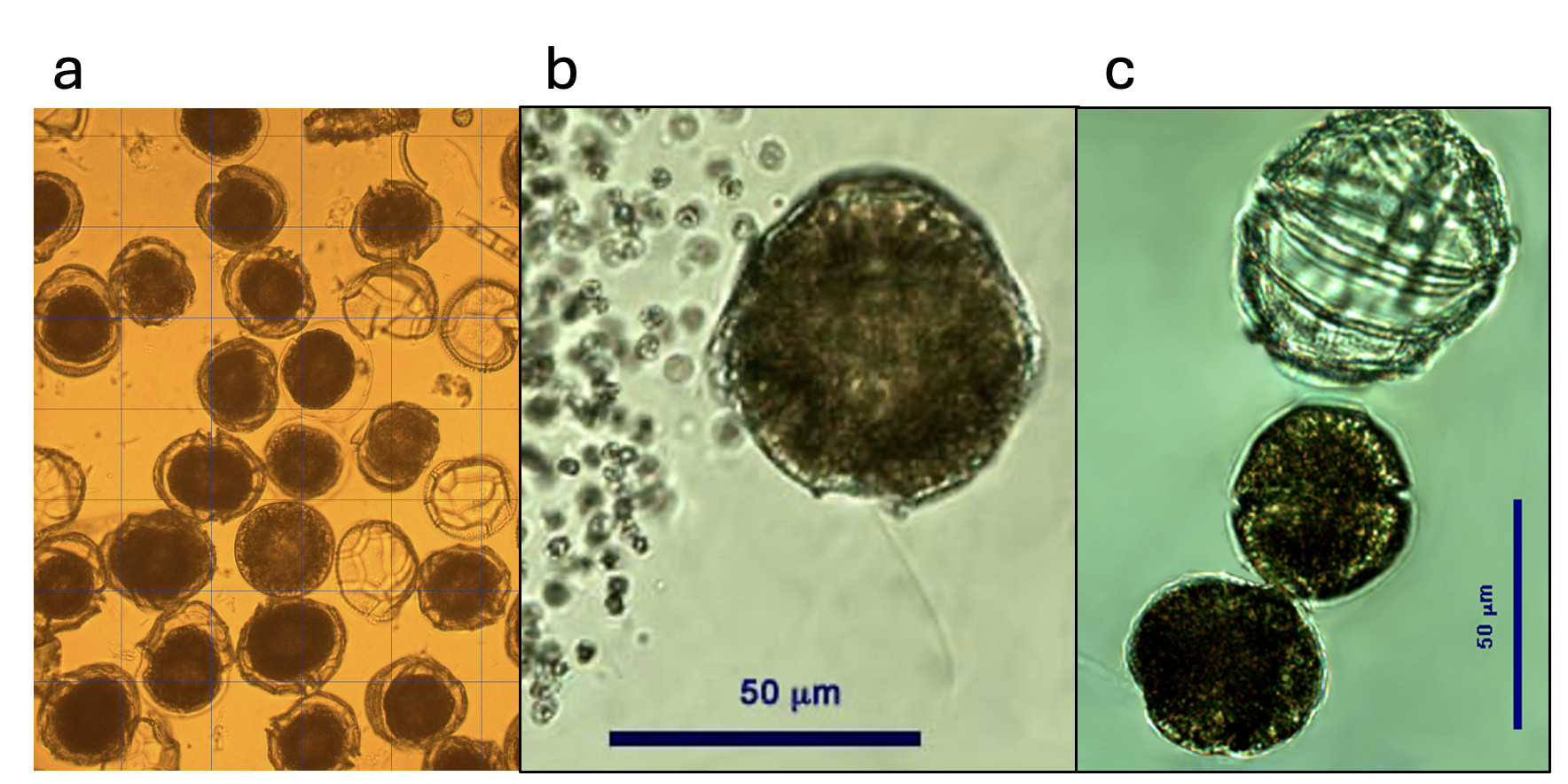
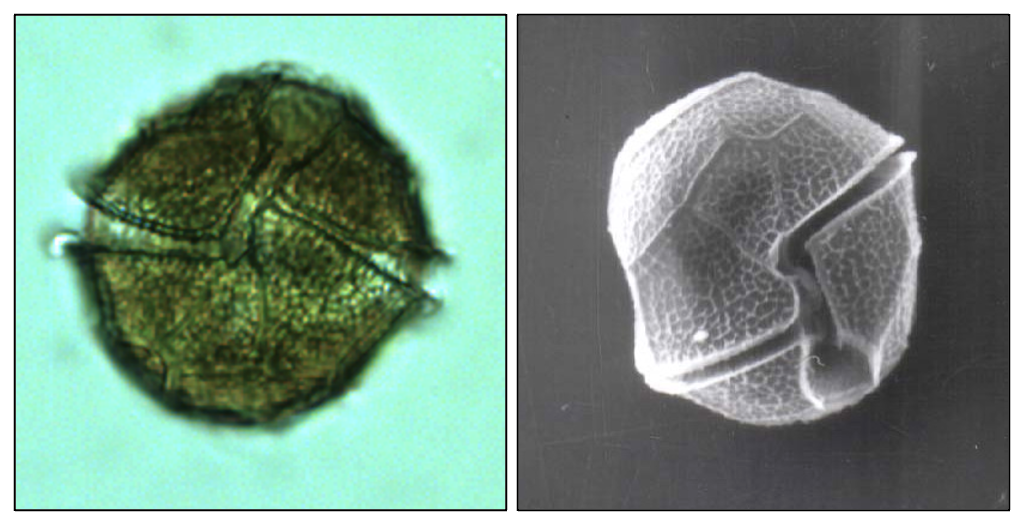
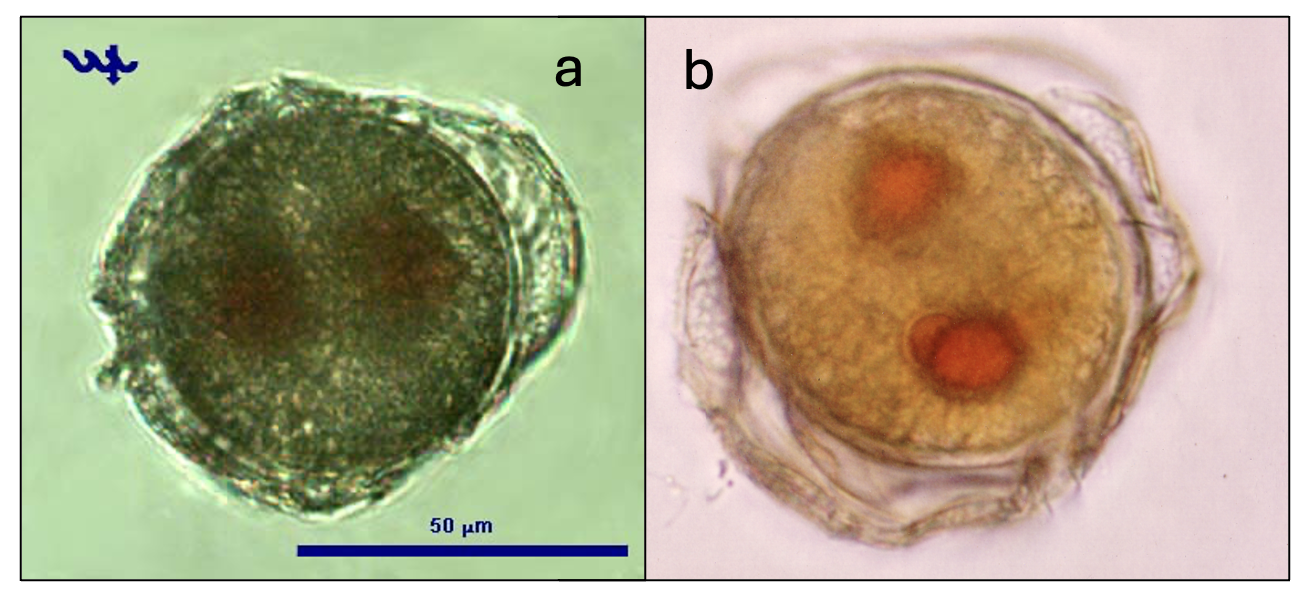
Ecology
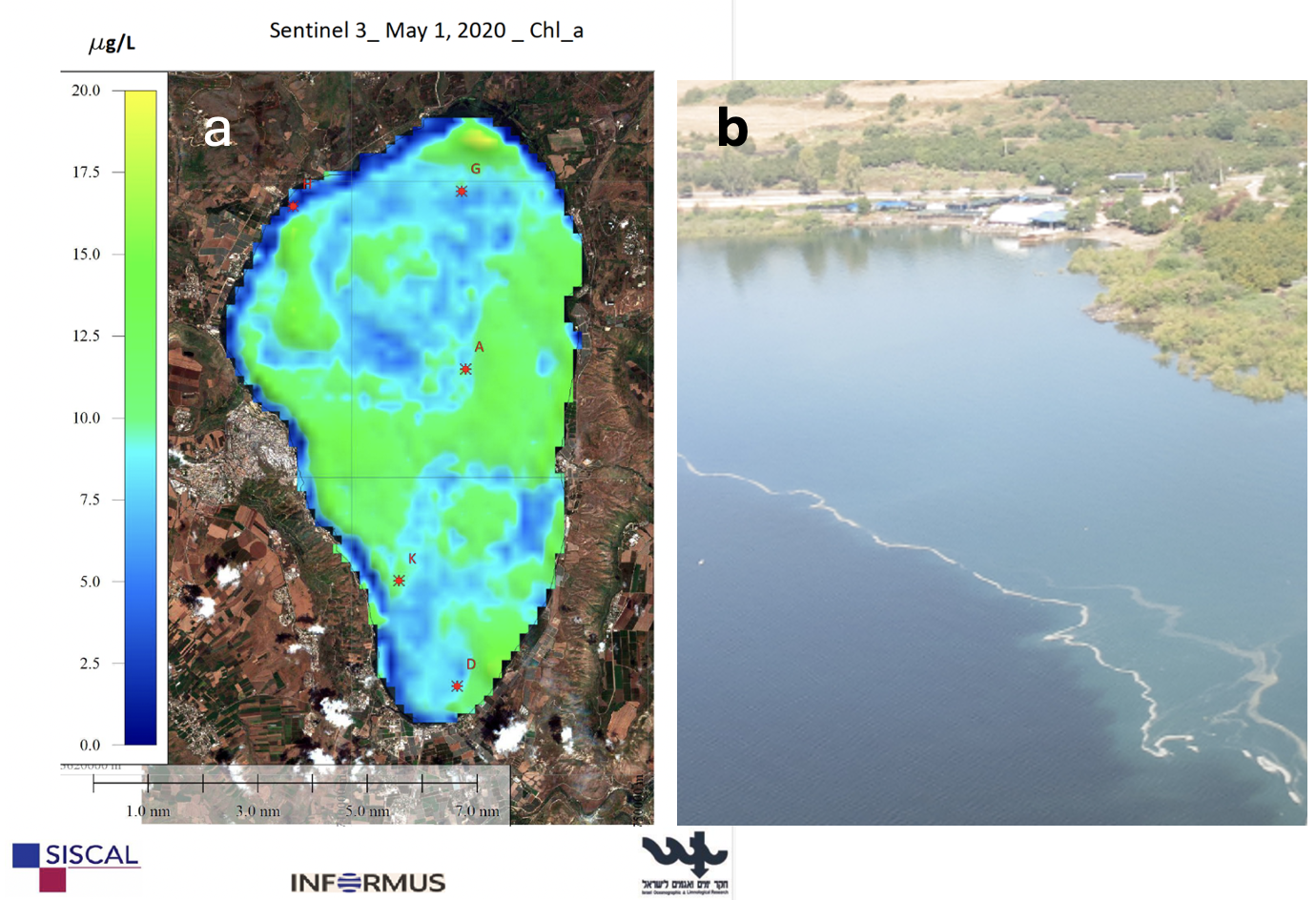
From the beginning of monitoring in Lake Kinneret in the 1960s, the regular late-winter-spring bloom of P. gatunense was the most salient feature of the Kinneret phytoplankton (Zohary et al. 2014). Those intense blooms could be observed as large patches of coffee-brown colored water (Plate 4), reaching up to several square kilometers in area. At the bloom peak P. gatunense reached mean epilimnion concentrations > 500 cells mL⁻¹ (Figs. 1, 2), or 150-250 g m⁻² of wet weight biomass and comprised >95% of total phytoplankton biomass. The blooms declined sharply in May-June (Fig. 2), shortly after water temperature exceeded 25ºC, thermal stratification was established, and nutrients became scarce, leaving behind resting cysts on the lake sediments. During the summer months P. gatunense concentrations in the water column were low, usually <5 cells mL⁻¹ (Fig. 2). Autumn turnover caused re-suspension of the cysts from the sediments, leading to their excystation and the initiation of the next year’s bloom. After many years of recorded constancy, in the mid-1990s, the regular pattern of late-winter spring P. gatunense blooms broke down, P. gatunense blooms have become irregular (Fig. 1) occurring only in high-rainfall years (Zohary et al. 2012), and in more recent years, not even in all of them. As in several other Kinneret dinoflagellates, cell size varies seasonally (Fig. 2), with largest sizes recorded in January-February, when nutrients are most plentiful, and smallest sizes in summer, after the bloom crash. In 2019 and 2020, when the lake’s water level rose after several years of being remarkably low and inundated for the first time shores that were overgrown by dense vegetation – exceptionally dense patches of P. gatunense (~1600 cells mL⁻¹) developed between flooded Tamarix trees (Zohary et al. 2026). Peridinium gatunense cells are too large to be grazed by crustacean zooplankton (Hambright et al. 2007). The native Cichlid fish feed primarily on Peridinium (Zohary et al. 1994) but overall, grazing has only a minor impact on the population. The primary loss of cells is due to death and decomposition in the water column. Second in importance is sinking to bottom sediments, where decomposition processes prevail but some grazing also takes place. An additional, less important losses is by parasitism by chytrid fungi in the water column (Alster and Zohary 2007).
Physiological features
Because Peridinium gatunense is a large cell, its growth rate is slow compared with that of other microalgae, with ambient doubling (turnover) time of ~7 days (Table 1). Lab experiments have shown maximal growth rates at 20-23 °C at light intensities ranging 50-200 μmol photons m-2 s-1. Growth rates decline at 25-27 °C (Berman and Dubinsky 1985). The specific photosynthetic rates (per unit chlorophyll) of P. gatunense are low (Table 1) in comparison with other phytoplankton prevailing in Lake Kinneret. But daily vertical migration and accumulation close to the surface in daytime (Berman and Rhode 1971) enhances photosynthesis. With darkness, the motile cells migrate down the water column to depths with higher nutrient concentrations. When CO2 levels become limiting (under intense bloom conditions), the dinoflagellates responded with higher levels of external and internal carbonic anhydrase activity and internal storage of inorganic C by means of a C concentrating mechanism (Berman-Frank et al. 1994). C content of P. gatunense, 45% of dry weight, is relatively high due to the thecae made of polyglucan, with practically no N, P. Allelopathic interactions between Peridinium gatunense and the bloom forming toxic cyanobacterium, Microcystis sp. were reported by Sukenik et al. (2002).
Table 1. Physiological parameters for Peridinium gatunense based on field measurements (from monitoring data using only dates when P. gatunense comprised > 90% of total phytoplankton biomass).
Table 1. Physiological parameters for Peridinium gatunense based on field measurements (from monitoring data using only dates when P. gatunense comprised > 90% of total phytoplankton biomass).
| Physiological parameter | Average ± SD | N | Reference |
|---|---|---|---|
| C:Chl ratio | 58.3 ± 19.3 | 243 | Yacobi and Zohary (2010) |
| Assimilation number (at depth of max photosynthesis), mg C mg Chl⁻¹ h⁻¹ | 1.56 ±0.80 | 36 | Yacobi and Zohary (2010) |
| Turnover time, d | 7.44±5.05 | 36 | Yacobi and Zohary (2010) |
| C content, % dry weight | 45.6±4.7 | 54 | J Erez lab, HUJ |
| N Content, % dry weight | 4.3±0.6 | 54 | J Erez lab, HUJ |
| C:P | 464±150 | Wynne et al. 1982 |
Environmental conditions
Peridinium gatunense biomass > 100 g m⁻² (considered a bloom) was associated with all incidences of Secchi depth < 1.5 m (Fig. 3) and TSS> 10 mg L⁻¹ (not shown), 70% of all incidences of turbidity > 4 NTU (not shown), 80% of all incidences with pH > 9.1 (Fig. 3), over 80% of the cases of excessive oxygen super-saturation (DO >13 mg L⁻¹, Fig. 3) and of TP> 0.05 mg L⁻¹, and over 60% of all cases where TN exceeded 1 mg L⁻¹, demonstrating how a single dominant species can modify the light conditions and chemical properties of its own environment. Factors that may have impacted P. gatunense biomass may have been salinity (no blooms at chloride> 280 mg L⁻¹) and availability of particulate organic N (Norg-part) for a mixotrophic species (Fig. 3). Its environmental preferences included temperature range of 16-23 °C, and probably particulate organic matter as a source of nutrients. This explains its proliferation in dense patches among inundated shore vegetation.
Additional figures
Cite this record as: Tamar Zohary, Alla Alster. 7 May 2026. Electronic publication. Israel Oceanographic & Limnological Research. https://kinneret-algae-atlas.org/ Searched on —.
Further reading
- Alster A, Zohary T (2007) Interactions between the bloom forming dinoflagellate Peridinium gatunense and the chytrid fungus Phlyctochytrium sp. Hydrobiologia 578:131-139.
- Alster A, Zohary T, Dubinsky Z (2006) Peridinium gatunense cyst type, abundance and germination in Lake Kinneret. Verh Internat Verein Limnol 29:2083-2086.
- Berman T, Dubinsky Z (1985) The autecology of Peridinium cinctum fa.westii from Lake Kinneret. Verh Internat Verein Limnol 22:2850-2854.
- Berman T, Rodhe W (1971) Distribution and migration of Peridinium in Lake Kinneret. Mitteilungen Internationale Vereinigung für Theoretische und Angewandte Limnologie 19:266-276.
- Berman T, Sherr BF, Sherr E, Wynne D, McCarthy JJ (1984) The characteristics of ammonium and nitrate uptake by phytoplankton in Lake Kinneret. Limnol Oceanogr 29:287-297.
- Berman T, Shteinman B (1998) Phytoplankton development and turbulent mixing in Lake Kinneret (1992-1996). J Plankton Res 20:709-726.
- Berman-Frank I, Erez J (1996) Inorganic carbon pools in the bloom-forming dinoflagellate Peridinium gatunense. Limnol Oceanogr 41:1780-1789.
- Berman-Frank I, Zohary T, Erez J, Dubinsky Z (1994) CO2 availability, carbonic anhydrase, and the annual dinoflagellate bloom in Lake Kinneret. Limnol Oceanogr 39:1822-1834.
- Boltovskoy A (1983) Peridinium cinctum f. westii del Mar de Galilea, sinonimo de Peridinium gatunense (Dinophyceae). Limnobios 2:413-418.
- Butow B, Wynne D, Sukenik A, Hadas O, Tel-Or E (1998) The synergistic effect of carbon concentration and high temperature on lipid peroxidation in Peridinium gatunense. J Phycol 20:355-369.
- Elgavish A, Elgavish GA, Halman M, Berman T (1980) Phosphorus utilization and storage in batch cultures of the dinoflagellate Peridinium cinctum fa. westii. J Phycol 16:626-633.
- Eren J (1969) Studies of development cycle of Peridinium cinctum fa. westii. Verh Internat Verein Limnol 17:1013-1016.
- Hader D-P, Liu S-M, Hader M, Ullrich W (1990) Photoorientation, motility and pigmentation in a freshwater Peridinium affected by ultraviolet radiation. Gen Physiol Biophys 9:361-371.
- Hambright KD, Zohary T, Gude H (2007) Microzooplankton dominate carbon flow and nutrient cycling in a warm subtropical freshwater lake. Limnol Oceanogr 52 (3):1018-1025.
- Hickel B, Pollingher U (1988) Identification of the bloom forming Peridinium from Lake Kinneret (Israel) as P. gatunense (Dinophyceae). Br Phycol J23:115-119.
- Hansen G, Flaim G (2007) Dinoflagellates of the Trentino Province, Italy. J Limnol. 66:107-141.
- Lindström K (1984) Effect of temperature, light and pH on growth, photosynthesis and respiration of the dinoflagellate Peridinium cinctum fa. westii in laboratory cultures. J Phycol 20:212-220.
- Lindström K (1991) Nutrient requirements of the dinoflagellate Peridinium gatunense. J Phycol 27:207-219.
- Liu S-M, Hader D-P, Ullrich W (1990) Photoorientation in the freshwater dinoflagellate, Peridinium gatunense Nygaard. FEMS Microbiol Let 73:91-101.
- McCarthy JJ, Wynne D, Berman T (1982) The uptake of dissolved nitrogenous nutrients by Lake Kinneret, Israel microplankton. Limnol Oceanogr 27:673-680.
- Messer G, Ben Shaul Y (1969) Fine structure of Peridinium westii, a freshwater dinoflagellate. J Protozool 15:272-280.
- Murik O, Kaplan A (2009) Paradoxically, prior acquisition of antioxidant activity enhances oxidative stress-induced cell death. Environ Microbiol 11 (9):2301-2309.
- Nevo Z, Sharon N (1969) The cell wall of Peridinium westii, a non cellilosic glucan. Biochim Biophys Acta 173:161-175.
- Pfiester LA (1977) Sexual reproduction of Peridinium gatunense (Dinophyceae). J Phycol 13:92-95.
- Pollingher U (1986) Phytoplankton periodicity in a subtropical lake (Lake Kinneret, Israel). Hydrobiologia 138:127-138.
- Pollingher U (1988) Freshwater armored dinoflagellates: growth, reproductive strategies and population dynamics. In: Sandgren C (ed) Growth and Reproduction Strategies of Freshwater Phytoplankton. Cambridge University Press, Cambridge, pp 134-174.
- Pollingher U, Hickel B (1991) Dinoflagellate associations in a subtropical lake (Lake Kinneret, Israel). Arch Hydrobiol 120:267-285.
- Pollingher U, Serruya C (1976) Phased division of Peridinium cinctum fa. westii (Dinophyceae) and development of the Lake Kinneret, Israel bloom. J Phycol 12:162-170.
- Pollingher U, Zemel E (1981) In situ and experimental evidence of the influence of turbulence on cell division processes of Peridinium cinctum fa.westii. (Lemm.) Lefevre. Br Phycol J 16:281-287.
- Rahat M (1968) Observations on the life cycle of Peridinium westii in a mixed culture. Isr J Bot 17:200-206.
- Rodhe W (1978) Peridinium cinctum fa. westii (Lemm.) Lef.: growth characteristics. In: Serruya C (ed) Lake Kinneret. Dr. Junk Publishers, The Hague, pp 275-283.
- Serruya C, Gophen M, Pollingher U (1980) Lake Kinneret: Carbon flow patterns and ecosystems management. Arch Hydrobiol 88:265-302.
- Sherr B, Sherr E, Berman T (1982) Decomposition of organic detritus: a selective role for microflagellate protozoa. Limnol Oceanogr 27:765-769.
- Spataru P, Zorn M (1976) Some aspects of natural food and feeding habits of Tilapia galilaea (Artedi) and Tiplapia aurea (Steindachner) in Lake Kinneret. Bamidgeh, The Isr J Aquaculture 28:12-17.
- Sukenik A, Eshkol R, Livne A, Hadas O, Rom M, Tchernov D, Vardi A, Kaplan A (2002) Inhibition of growth and photosynthesis of the dinoflagellate Peridinium gatunense by Microcystis sp.(cyanobacteria): a novel allelopathic mechanism. Limnol Oceanogr 47:1656-1663.
- Usvyatsov S, Zohary T (2006) Lake Kinneret continuous time-depth chlorophyll record highlights major phytoplankton events. Verh Internat Verein Limnol 29:1131-1134.
- Vardi A, Berman-Frank I, Rozenberg T, Hadas O, Kaplan A, Levine A (1999) Programmed cell death of the dinoflagellate Peridinium gatunense is mediated by CO2 limitation and oxidative stress. Current Biol 9:1061-1064.
- Vardi A, Eisenstadt D, Murik O, Berman-Frank I, Zohary T, Levine A, Kaplan A (2007) Synchronization of cell death in a dinoflagellate population is mediated by an excreted thiol protease. Environ Microbiol 9:360-369.
- Vardi A, Schatz D, Beeri K, Motro U, Sukenik A, Levine A, Kaplan A (2002) Dinoflagellate-cyanobacterium communication may determine the composition of phytoplankton assemblage in a mesotrophic lake. Current Biol 12:1767-1772.
- Wynne D (1981) Phosphorus, phosphatases and the Peridinium bloom in Lake Kinneret. Verh Internat Verein Limnol 21:523-527.
- Wynne D, Patni NJ, Aaronson S, Berman T (1982) The relationship between nutrient status and chemical composition of Peridinium cinctum during the bloom in Lake Kinneret. J Plankton Res 4:125-136.
- Yacobi YZ, Pollingher U, Gonen Y, Gerhard V, Sukenik A (1996) HPLC analysis of phytoplankton pigments from Lake Kinneret with special reference to the bloom-forming dinoflagellate Peridinium gatunense (Diniophyceae) and chlorophyll degradation products. J Plankton Res 18:1781-1796.
- Yacobi YZ, Zohary T (2010) Carbon:chlorophyll a ratio, assimilation numbers and turnover times of Lake Kinneret phytoplankton. Hydrobiologia 639:185-196.
- Zohary T (2004) Changes to the phytoplankton assemblage of Lake Kinneret after decades of a predictable, repetitive pattern. Freshwat Biol 49:1355-1371.
- Zohary T, Erez J, Gophen M, Berman-Frank I, Stiller M (1994) Seasonality of stable carbon isotopes with Lake Kinneret pelagic food web. Limnol Oceanogr 39:1030-1043.
- Zohary T, Hadas O, Pollingher U, Kaplan B, Pinkas R, Güde H (2000) The effect of nutrients (N, P) on the decomposition of Peridinium gatunense cells and thecae. Limnol Oceanogr 45:123-130.
- Zohary T, Nishri A, Sukenik A (2012) Present-absent: a chronicle of the dinoflagellate Peridinium gatunense from Lake Kinneret. Hydrobiologia 698:161-174.
- Zohary T, Pollingher U, Hadas O, Hambright KD (1998) Bloom dynamics and sedimentation of Peridinium gatunense in Lake Kinneret. Limnol Oceanogr 43:175-186.
- Zohary T, Sukenik A, Berman T (2014). Peridinium gatunense. Chap. 11 In: Zohary T, Sukenik A, Berman T, Nishri A. [eds] Lake Kinneret: Ecology and Management, pp 191-212. Springer, Heidelberg.
- Zohary T, Alster A, Cummings D, Dolev A, Eckert W, Gal G, Gasith A, Guk E, Leibovici E, Ofir E, Varulker S, Be’eri-Shlevin Y. 2026. Inundated Tamarix forest – a novel, climate change-induced littoral habitat in Lake Kinneret. Inland Waters 16(1), 2557696.